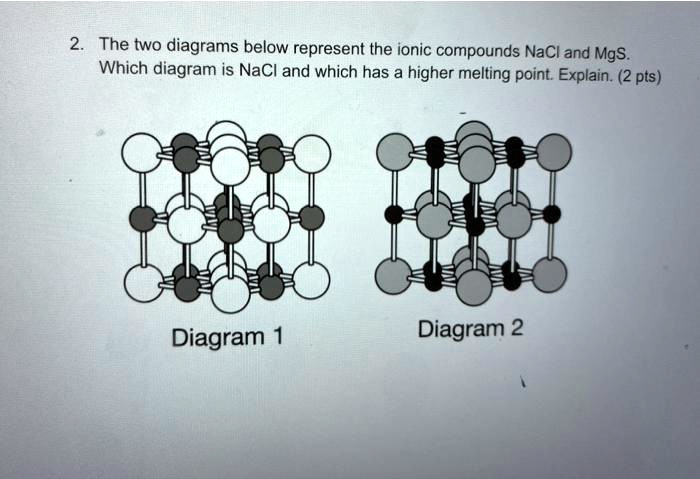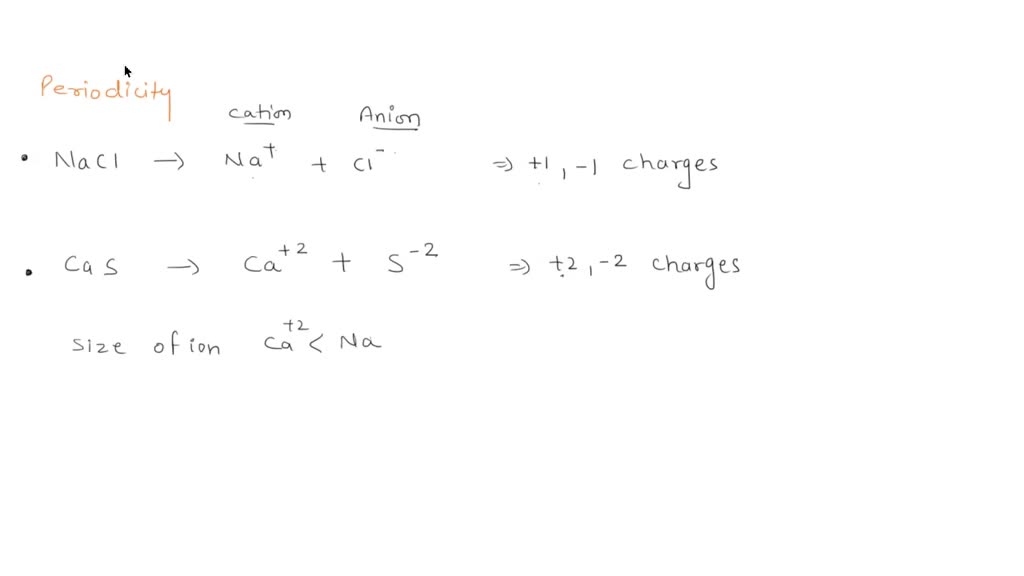
SOLVED: between NaCl and CaS, which do you think has a higher melting point? use the concepts of periodic trends and ionic bonding to justify your answer

A SEM picture of porous HA after the removal of NaCl and PVA as pore... | Download Scientific Diagram

Comparison of the contact angle variation between NaCl and KCl solution... | Download Scientific Diagram

RHEED images of (1) initial and (2) final GaAs deposition on thick NaCl... | Download Scientific Diagram

Effect of NaCl and KCl on the Solubility of Amino Acids in Aqueous Solutions at 298.2 K: Measurements and Modeling | Industrial & Engineering Chemistry Research

Effect of KCl, NaCl and CaCl2 mixture on volume combustion synthesis of TiB2 nanoparticles - ScienceDirect
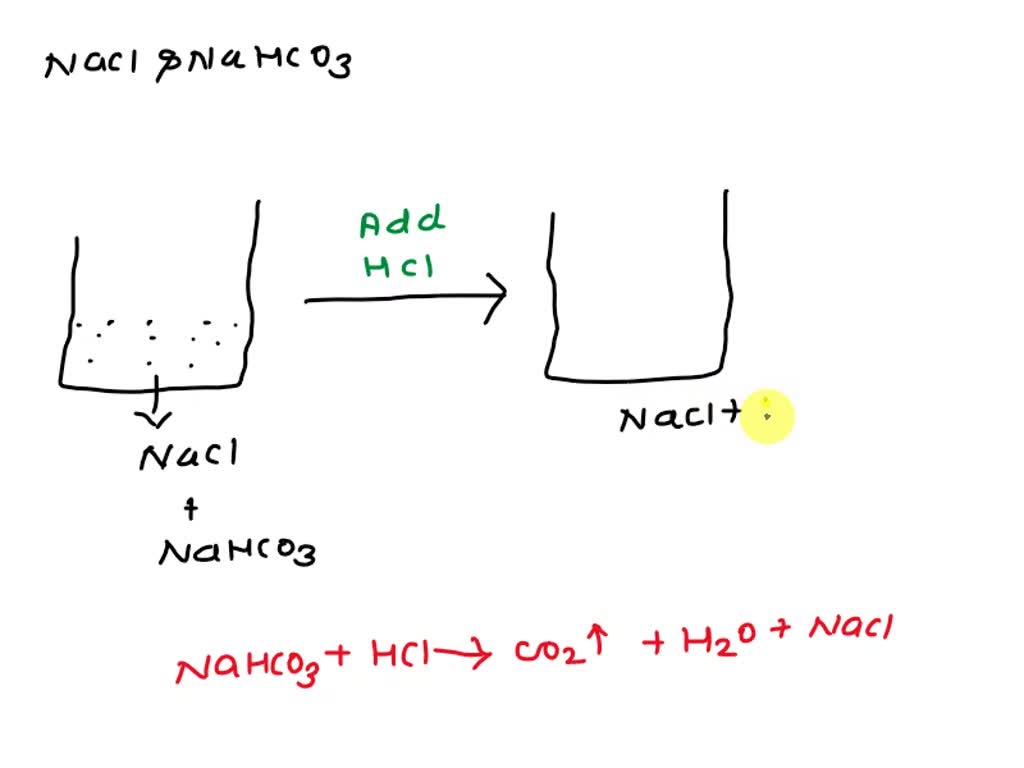
SOLVED: What is the best way for separation of a mixture NaCl and NaHCO3 in 200 mL water? HCl is available and we need to separate NaCl and water in two different

Effect of NaCl and KCl on the Solubility of Amino Acids in Aqueous Solutions at 298.2 K: Measurements and Modeling | Industrial & Engineering Chemistry Research

SOLVED: Suppose you have a mixture of sodium chloride, NaCl, and carbon, C. Explain how you can use water to separate the two substances.

a) Fabrication of Au dewetted nano-particles (nano-caps) on NaCl by... | Download Scientific Diagram

NaCl and KCl mediate log increase in AAV vector particles and infectious titers in a specific/timely manner with the HSV platform - ScienceDirect
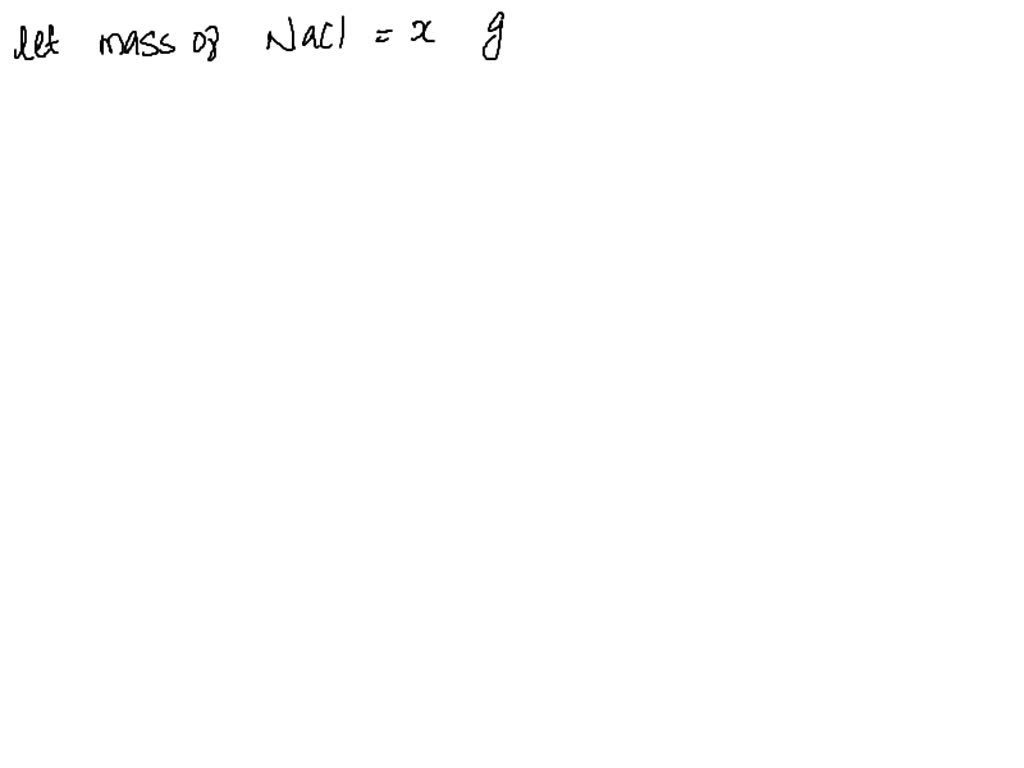
SOLVED: A 0.1824-g sample that contains both NaCl and NaBr is titrated with 0.1052 M AgNO3, requiring 26.48 mL. Calculate the percentages of NaCl and NaBr in the sample. NaCl = 58.442 NaBr = 102.89

CFMEU hopes preferencing Labor last will send message to State Government over New Acland near Oakey, west of Toowoomba | The Chronicle
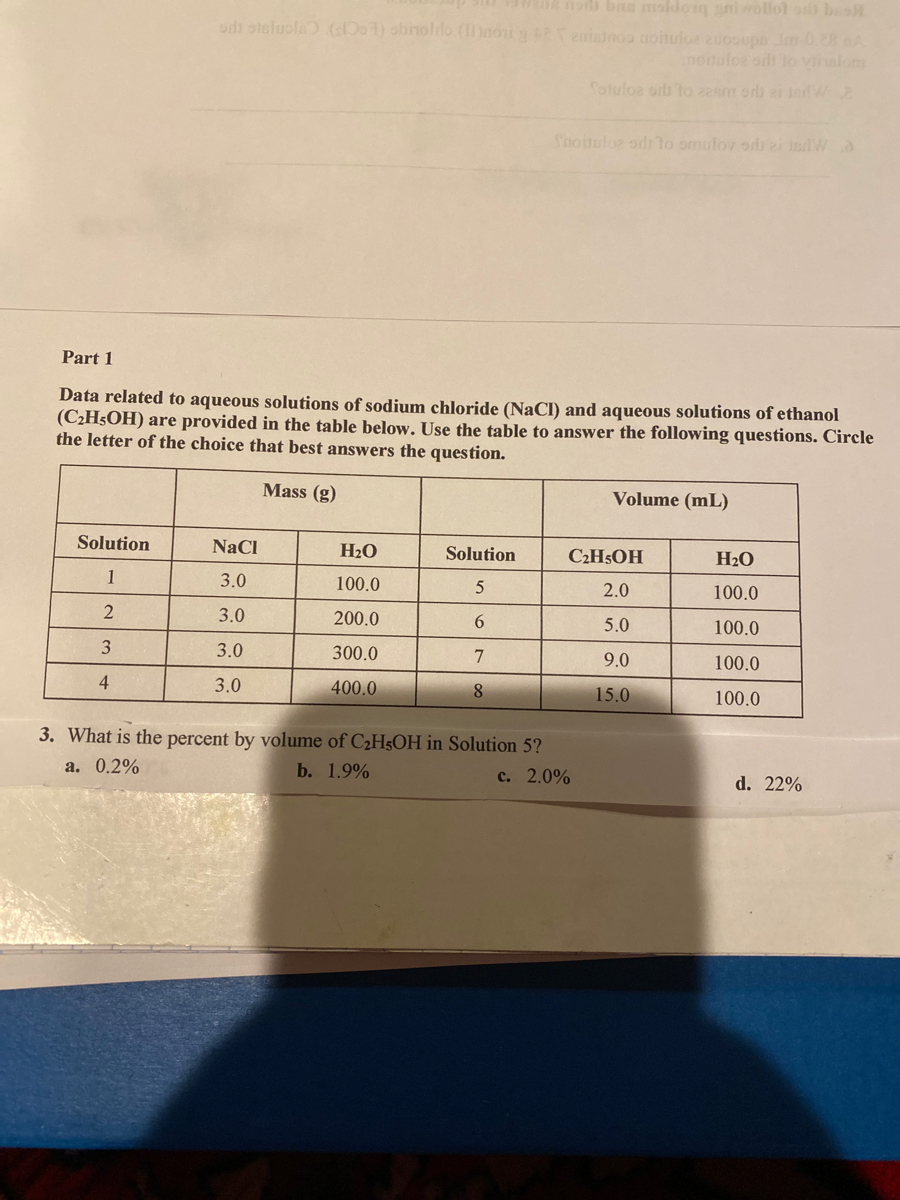
![SOLVED: 10.0 g of NaCl and 100.0 g of Select ] HzO 50.0 mL of ethanol, CzHsOH and Water is the solute and ethan 10.0 mL of HzO 0.20 Lof 02 and SOLVED: 10.0 g of NaCl and 100.0 g of Select ] HzO 50.0 mL of ethanol, CzHsOH and Water is the solute and ethan 10.0 mL of HzO 0.20 Lof 02 and](https://cdn.numerade.com/ask_images/4fa281ca316d40fda76e19c1f8aca2f8.jpg)
SOLVED: 10.0 g of NaCl and 100.0 g of Select ] HzO 50.0 mL of ethanol, CzHsOH and Water is the solute and ethan 10.0 mL of HzO 0.20 Lof 02 and

Comparison of the different contribution to the slip velocity (36) for... | Download Scientific Diagram

SOLVED: If you start with mixture of NaCl and KCIO3 that weighs 3.00 g and the mass of the sample decreases by 0.75 g after heating, what is the best explanation for

Dissolution of sandstone powder and thin slice in the 3 m NaCl and 1 m... | Download Scientific Diagram
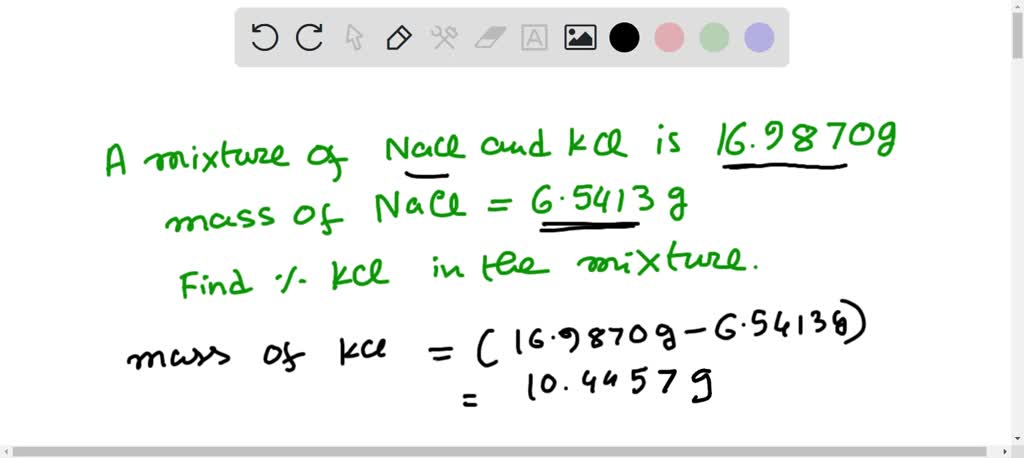
SOLVED: Suppose 16.9870 g sample of a NaCl and KCl mixture contains 6.5413 of NaCl What is the percent of KCl in the mixture?
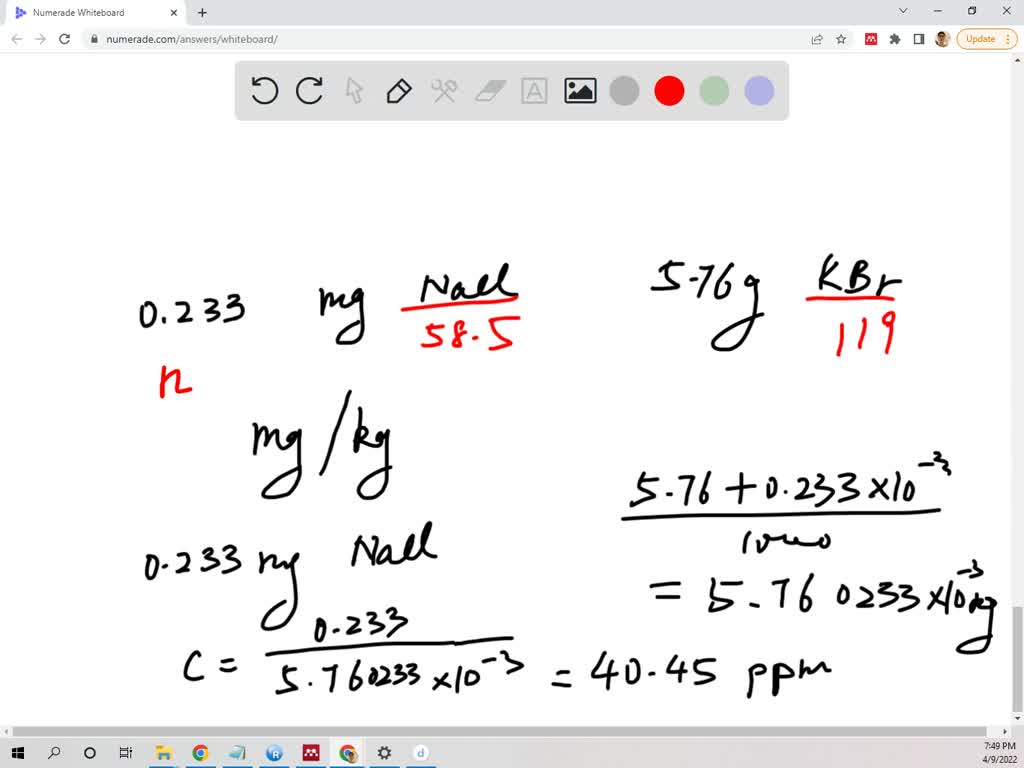
SOLVED: Consider mixing 0.233 milligrams of sodium chloride (NaCl) and 5.76 grams of potassium bromide (KBr) until the mixture appears as a uniform powder. What is the concentration of sodium chloride in
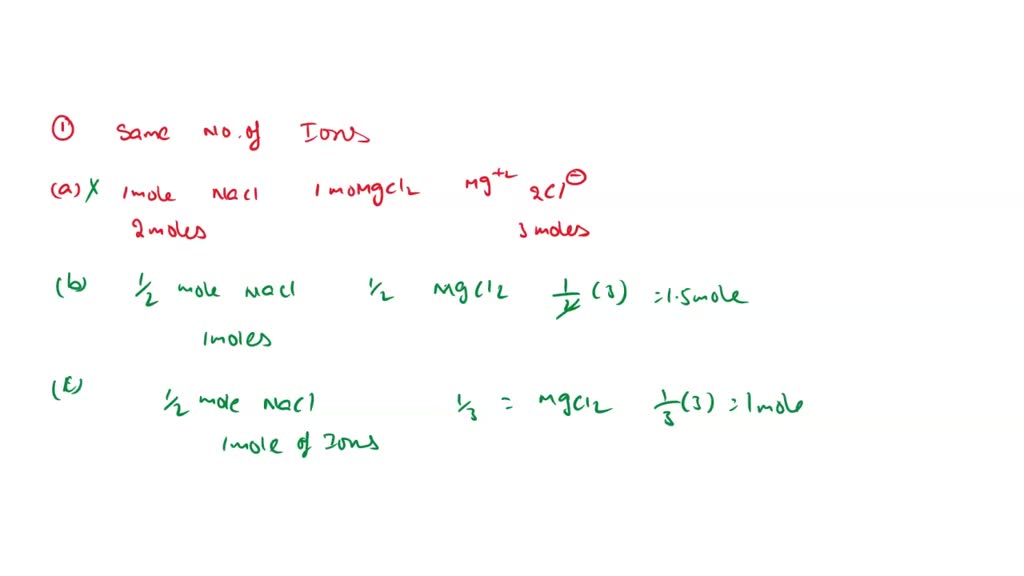
SOLVED: Whi(a) I mole of NaCl and I mole of MgCl2(b) 1/2 mole of NaCl and 1/2 mole of MgCl2(c) 1/2 mole of NaCl and 1/3 mole of MgCl2(d) 1/3 mole of